Floy × Scarlet
3 AI devices MDR certified – plus a completed ISO transfer & scope extension – all in 4 months
How Floy got a suite of innovative SaMD approved for the European market
"Scarlet have a very strong understanding of AI medical devices and their regulatory context, it made working with them for our devices very straightforward."
Dr Lea-Elena Braunschneider, Medical Excellence Lead, Floy
The Challenge
Floy wanted to complete multiple regulatory workstreams for a collection of radiology products, and needed a Notified Body with clear AI expertise
Customer Snapshot
- Innovative, Germany-based AI start-up
- Developed a range of AI-powered medical devices supporting radiologists
- Looking for a Notified Body with deep knowledge of cutting-edge AI for EU MDR & ISO 13485 certification
Product Impact
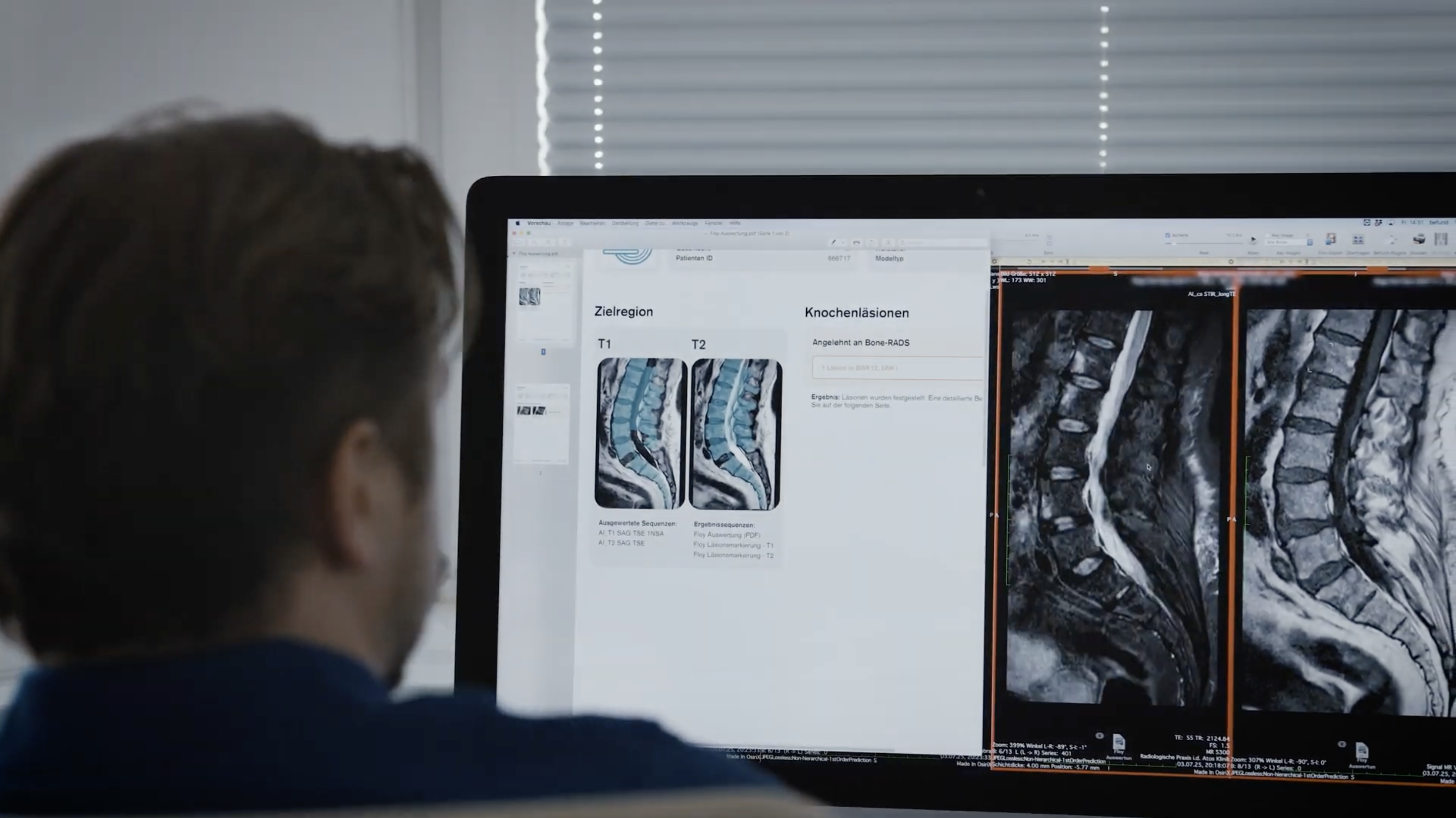
- Augmenting scans with bone-mineral-density scores and enhanced aneurysm detection
The Scarlet Process
Scarlet's intelligent account management allowed Floy to release multiple AI-driven medical devices
How It Worked
To ensure an efficient process, we:
- Used Scarlet’s AI specialists to efficiently understand Floy’s products – avoiding needless back and forth
- Provided expert regulatory feedback – avoiding inefficient internal efforts for Floy
- Maintained regular, transparent contact – avoiding uncertainty over timelines
The Results
Scarlet’s approach resulted in:
- EU MDR certification for three cutting-edge devices
- A completed technical-file assessment in just 6 weeks
- A seamless ISO-certificate transfer and scope extension
- Floy is set up to bring many more innovative devices to the market
"Even with several regulatory projects running in parallel, Scarlet's customer support meant that everything was really well coordinated and easy to manage.”
Dr Lea-Elena Braunschneider, Medical Excellence Lead, Floy
Need to certify an AI medical device/SaMD?
Schedule a call or demo, or simply get more info about the Scarlet process and platform.
