Augmedit × Scarlet
EU MDR & ISO 13485 certification for multiple Augmented Reality products
How Augmedit got their innovative SaMD certified for the European market
“Working with Scarlet actually led to us creating higher quality SaMD.
Their expert feedback meant we refined our work and built better versions of our products.”
Claartje Ypma, Co-founder & CEO, Augmedit
The Challenge
Augmedit needed a Notified Body with SaMD expertise to understand and certify their ground-breaking devices
Customer Snapshot
- Netherlands-based Augmented Reality (AR) & Extended Reality (XR) start-up
- Developed a suite of solutions addressing neurosurgery navigation and planning challenges
- Looking for a Notified Body with deep knowledge of cutting-edge SaMD, to gain EU MDR & ISO 13485 certification, and facilitate market access & product improvements
Product Impact
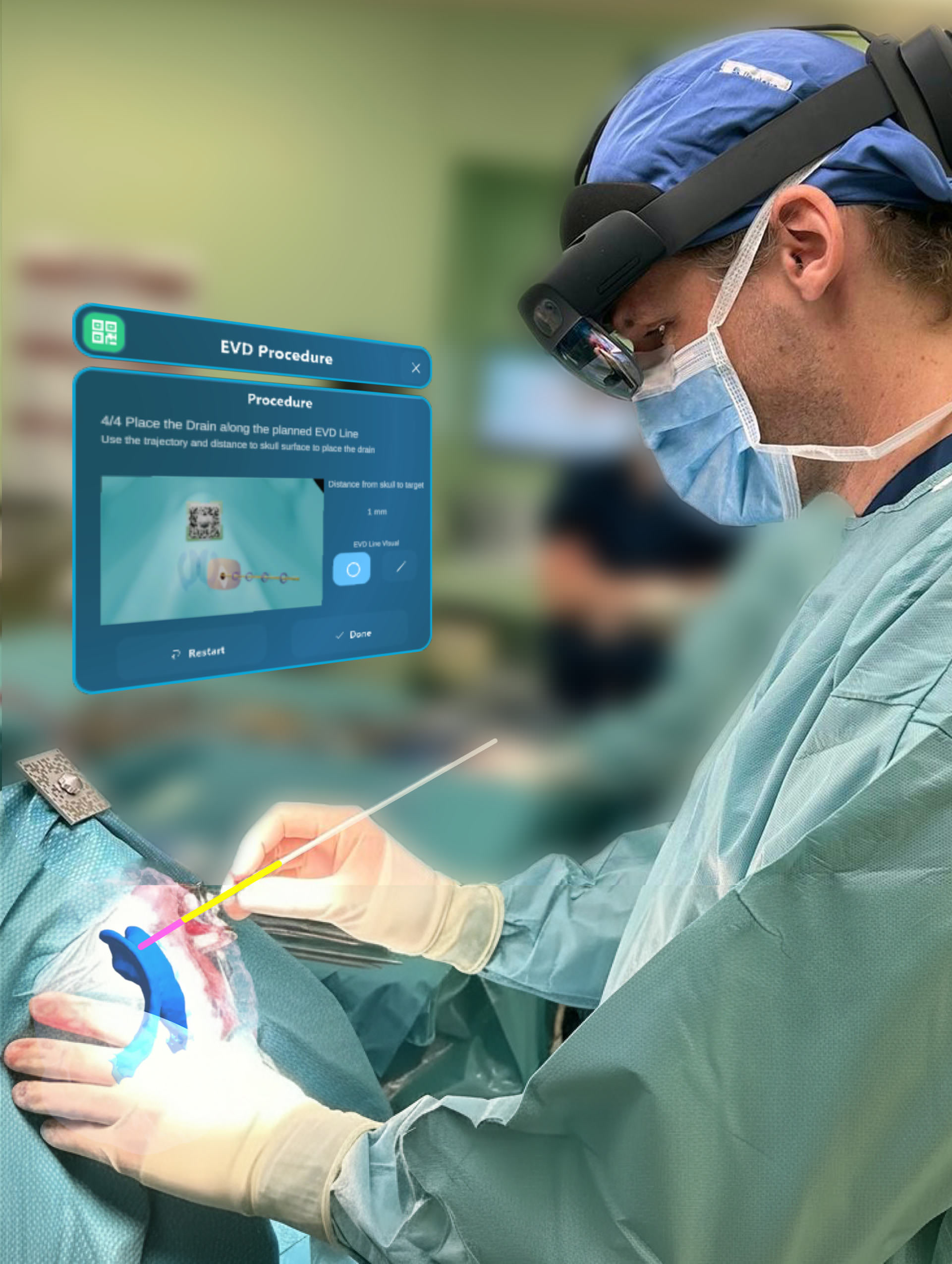
- Bringing AI-driven AR SaMD to surgery, to improve real-time anatomical understanding and surgical outcomes
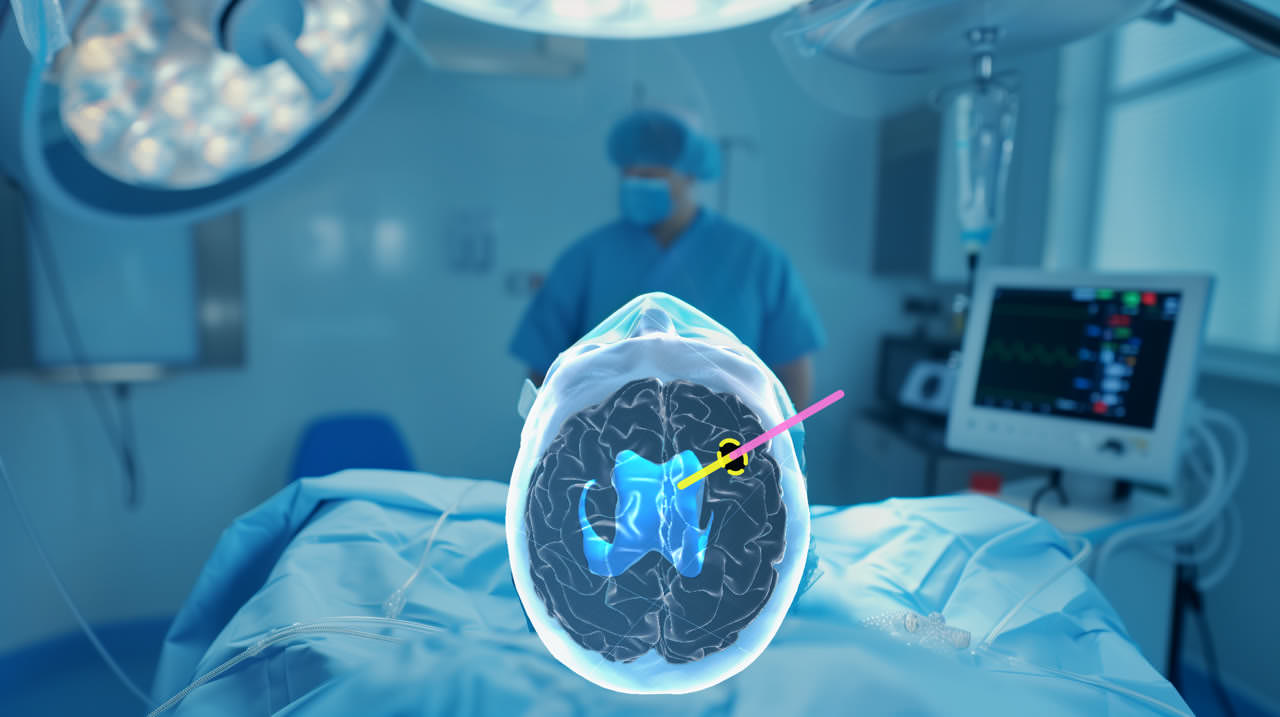
- Already seen its first use with patients: EVD (external ventricular drain) procedures performed using Augmedit’s AR, with a patient-specific hologram of the brain
The Scarlet Process
Scarlet’s SaMD expertise and high-touch account management helped Augmedit to get the very best versions of their technology to doctors
How It Worked
To ensure an efficient process, we:
- Used Scarlet’s AI specialists to efficiently understand Augmedit’s products – avoiding needless back and forth
- Provided expert regulatory feedback – avoiding inefficient internal efforts for Augmedit
- Maintained regular contact and responsive support to Augmedit – avoiding uncertainty over timelines and planning
The Results
Scarlet’s approach resulted in:
- EU MDR certification for three cutting-edge devices
- Augmedit can now expand and efficiently iterate their ground-breaking technology
- Products now in use with first patients in the Dutch healthcare system
“It’s great to work with a Notified Body that truly understands AI.”
Claartje Ypma, Co-founder & CEO, Augmedit
Need to certify an AI medical device/SaMD?
Schedule a call or demo, or simply get more info about the Scarlet process and platform.
