How to use Secure Data Environments to deliver PMCF for SaMD
Post-market clinical follow-up (PMCF) is an ongoing process to update the clinical evaluation over the device lifetime. To meet the requirements of EU MDR Annex XIV, it needs to be proactive and systematic, so that it confirms the safety and performance of the device under conditions of normal use.
This presents several challenges to manufacturers:
- Data gathering: Medical record systems are fragmented, often inaccessible to the manufacturer outside of a clinical study
- Clinical trials: Frequently resource intensive
- Commercial plans: Sales roadmaps are not usually aligned with evidence-generation requirements
How can SDEs help?
Secure Data Environments (SDEs) offer a secure and sustainable way to gather and robustly examine real-world data (RWD) at scale. In accordance with MDCG 2020-7, it’s not the presence of RWD that matters to Notified Bodies, but how it’s used. Real-world evidence (RWE) is acceptable when the dataset is well characterised, the analysis is pre-specified, and the association with clinical claims is justified. SDEs can support this by providing access to structured, longitudinal patient data, collected during routine care (i.e. normal conditions of use).
What exactly is an SDE?
A Secure Data Environment (SDE) refers to the technical and governance infrastructure that allows researchers to access sensitive health data remotely, without moving or extracting it. It provides a secure, auditable environment for analysis, typically used for working with RWD that is too sensitive to share openly.
While SDEs often host large-scale, linked datasets such as electronic health records or registries, the environment (SDE) and the data (RWD) are distinct, and both must be robust for the resulting evidence to be acceptable to Notified Bodies.
In the UK's NHS, data from GP clinics, hospitals, manufacturers, registries, and more can be connected and examined in one place. Unlike setting up a new multicentre study from scratch, an SDE allows you to plug into existing RWD ethically and securely.
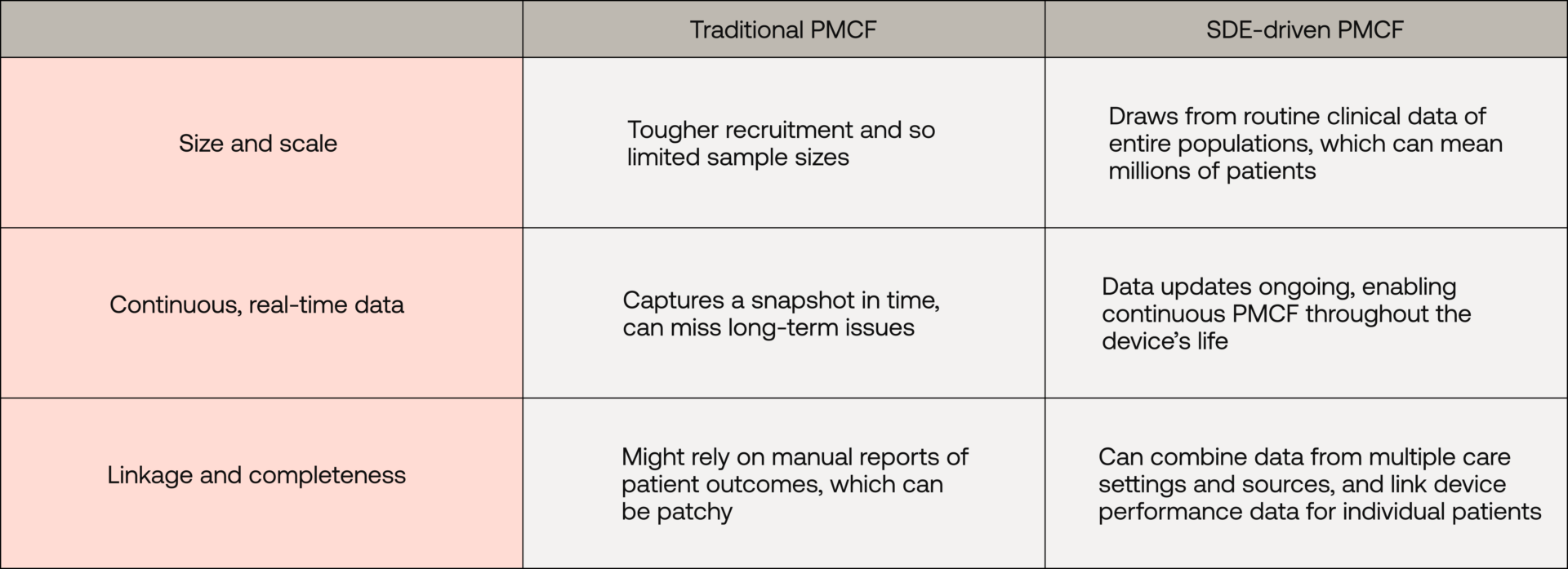
SDEs vs. traditional data collection
How is an SDE-driven approach different from traditional post-market studies or registries? Let’s compare:
The TRICORDER example
The London SDE is coming online, storing linked clinical, cost (health-economic), pharmacy, mental health and social care data across both primary and secondary care. It covers millions of patients with unique urban/rural, socioeconomic, ethnic and health diversity - enabling post-market evidence generation that is internationally generalisable.
One recent UK project gives a glimpse of the potential for SDE-driven PMCF. The TRICORDER trial – a large NHS study led by Imperial College London and involving Senior Clinical Scientists at Scarlet – set out to independently evaluate the post-market impact of a Class IIa CE-marked device for detection of heart disease in routine primary care.
The project spanned 205 GP practices and over 1.5 million patients, with outcomes of this randomised controlled trial examined in the London SDE. For manufacturers, this represents a potential step-change in the feasibility of large-scale, scientifically robust, highly efficient PMCF through SDEs to glean powerful evidence on how their device performs in the wild.
Tackling PMCF’s biggest challenges
How do SDEs specifically help solve three key PMCF problems of disconnected evidence, low recruitment, and biased data?
Connecting the evidence pipeline: Post-market evidence is often mistakenly considered a ‘bolt-on’, separate from pre-market trials and not easily updated. SDEs address this by enabling a continuous evidence pipeline. Data flows in from real-world use and can be analysed alongside or in light of earlier clinical trial results.
For instance, an SDE-based registry might recruit an uncapped number of patients and run indefinitely as long as the device is on the market, in stark contrast to a typical 6-12 month study that ends as soon as it meets its enrolment target. This means evidence is always up to date and reflective of current practice.
Supercharging recruitment (without “recruiting”): Traditional post-market trials often face low enrolment. It’s hard (and expensive) to get many hospitals and patients on board for additional research once a product is already available.
SDEs offer a clever workaround: you’re essentially leveraging data from patients who are already using/exposed to the device as part of normal care. There’s no giant advertising campaign or lengthy consent process needed to find participants from scratch. The data accumulates organically as the device is used.
Reducing bias: SDE-based PMCF can substantially mitigate biases traditionally found in observational studies. Notified Bodies expect evidence that supports valid causal inference or strong association. This requires clarity on population definition, outcome capture, and control of confounders.
SDEs allow precise cohort identification that can represent a wider population (the NHS “million” rather than the select “few hundred” in a trial), including community-care settings and diverse patient demographics. That means analyses can include older patients, multiple comorbidities, and others often excluded from trials, leading to findings that are more generalisable. Also, SDEs don’t cherry-pick sites; data comes from all types of clinical sites, not just centres of excellence. This avoids the bias of only seeing results from top-tier institutions that might get better outcomes. While observational data will always require careful analysis to adjust for confounding factors, having such a rich, large dataset helps statisticians use robust methods to control for those factors.
Informing commercial strategy
Data gathered via an SDE-based PMCF can play a dual role in strengthening the case when seeking national commissioning, insurance coverage, or hospital adoption.
Going into a health-technology assessment meeting not just with a small pre-market trial, but with a rich dataset showing how the device performed across thousands of NHS patients in routine care is compelling.
For SaMD manufacturers, SDEs offers a practical way to generate robust, real-world evidence at scale. This means strengthening clinical claims, supporting regulatory pathways, demonstrating value in routine care, and demonstrating impact across diverse patient populations.

Want Scarlet news in your inbox?
Sign up to receive updates from Scarlet, including our newsletter containing blog posts, sent straight to you by email.


